Modified from http://silverfalls.k12.or.us/staff/read_shari/Alien%20Periodic%20Table.pdf
Alien Periodic Table
Periodic Law is an important concept to understand. Over several centuries, multiple scientists have helped us understand the nature of matter and atoms by making contributions to the organization of the modern Periodic Table. This activity is designed to help you read information and organize that information based on known trends and patterns from the Periodic Table. As you go through this exercise, pay attention to the clues to think about how each “alien” atom relates to a real-world “Earth” element.
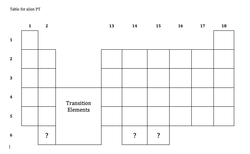
Procedure: You are a part of a collection of scientists who have been chosen to assist a group of alien scientists. In order to be able to converse scientifically, you must learn their language, and most importantly, you must arrange their elements according to the trends that exist in the periodic table. Below are clues for the alien's elements. So far, the aliens have only discovered elements in groups 1, 2, and 13-18, and periods 1-5. Although the names of the elements are different, they must correspond to our elements if our belief of universal elements holds true. Read each clue carefully, and then place the symbol for that clue's element in the blank periodic table provided.
Clues:
- Livium (Lv): This element is responsible for life. It has 2 electron energy levels and 4 electrons available for bonding in the outermost energy level.
- Computerchipium (Cc): This element is important for its use as a semiconductor in computers.
- Lightium (L): This is the lightest of elements; aliens used to use it in their aircraft until their aircraft caught fire in a horrific accident.
- Breathium (Br): When combined with Lightium (L), it makes the alien's most common liquid whose formula is L2Br.
- Frenchchemium (F): This element is a metal found in period 4 group 13.
- Moonium (Mo): An element with an atomic number of 34.
- Explodium (Ex): This element is the most reactive metal on the alien's table.
- Violetium (V): This element is found in bananas. When burned, it has a violet colored flame.
- Sparkium (Sp) and Burnium (Bu) are members of the alkali metal group, along with Violetium(V) and Explodium (Ex). Their reactivity, from least to greatest, is Sp, Bu, V, Ex.
- Balloonium (Ba): A noble gas used to fill balloons
- Toothium (To): This element is added to juices to help build strong bones and teeth.
- Metalloidium (Md) and Poisonium (Po): These two elements are metalloids found in period 4. Po is the more massive.
- Lowigium (Lo): A period 4 halogen
- Darkbluium (Dk): A radioactive isotope (electron capture) of this element has a mass number of 115.
- Hugium (Hu): The element on the alien's periodic table that has the most mass.
- Glucinium (Gl): The element found in period 2, group 2.
- Reactinium (Re): The most reactive non-metal on the periodic table.
- Balloonium (Ba), Signium(Si), Stableium(Sb), Supermanium (Sm), and Hugium (Hu) are all noble gases. They are arranged above from least to most massive.
- Burnium (Bu) has one valence electron. It is found in most foods. When burned, it has an orange colored flame.
- Cannium (Cn): This element helps to preserve foods; it is used in can manufacturing.
- Blue-whitium (Bw), Bauxitium (Xi), Computerchipium (Cc), Bringer-of-lightium (Bl), Stinkium (Sk), Purium (P), and Stableium (Sb) are all found in period 3. Bw has 2 electrons in its outer energy level, Xi has 3, Cc has 4, Bl has 5, Sk has 6, P has 7 and Sb has 8.
- Scottishium (Sc): A metal element found in group 2.
- Infectium (If): This element has been used as a dietary supplement and astringent in eye wash.
- Abundantcium (Ab): One of the most abundant gases in the universe, Ab has 7 protons, 7 neutrons, and 7 electrons.
- Some additional clues: The number after the symbol indicates the number of electrons in the outer energy level: Notalonium (Na, 5); Earthium (E; 6); Batterium (B; 3).
Post Activity Analysis Questions:
- Use the information provided in the clues to develop a description for REACTIVITY. Relate reactivity to metallic character, ionization energy, electronegativity and electron affinity.
- What do you think is the relationship between atomic mass and atomic number? Develop a quantitative expression that approximates the relationship.
- Describe a trend in metallic character. How does this relate to the metalloids? What connections can be made to reactivity?
- What Earth element is represented as Ab? What is the relative composition of “abundantcium” in air?
- Bauxite is an ore (valuable natural resource) that can be mined. Use the clues to determine which element can be extracted from bauxite? Explain your choice.
- Alien scientists know that there are at least three more elements (indicated by question marks on their chart). One of the unknown metals has already been named because of its characteristics, Metalgravitium (Mg), a heavy metal with no known benefits to the human body. Using the format that exists in the other clues, develop clues for the remaining two unknown elements. Propose a symbol and name for the two remaining elements. Develop an explanation for why the element is called metalgravitium.